ACPN 宣布发布《自然健康产品循证功效体系技术标准》
ACPN Announces the Publication of the Technical Standard for Evidence-based Efficacy System of Natural Health Products
ACPN 宣布发布《自然健康产品循证功效体系技术标准》
Toronto, Canada — July 2025 – The Association of Certified Professional Nutritionists (ACPN) proudly announces the official publication of the Technical Standard for Evidence-based Efficacy System of Natural Health Products (自然健康产品的循证功效体系技术标准). This landmark document establishes the first comprehensive, multi-platform technical guideline designed to confirm and standardize the efficacy of natural health products. This initiative represents a significant step forward for the nutrition and natural health industry in Canada and beyond, aligning with ACPN’s mission to promote science-based health practices.
加拿大多伦多 — 2025年7月 —— 加拿大注册执业营养师公会(ACPN)隆重宣布,正式发布《自然健康产品循证功效体系技术标准》。这一里程碑式文件,首次建立了一个全面、多平台的技术指南,用于确认和规范自然健康产品的功效。此举标志着加拿大乃至全球营养与自然健康产业迈出了重要一步,与ACPN推动科学化健康实践的使命高度契合。
The Technical Standard was developed through collaboration between leading scientific organizations, including Keypoint Boston LLC, the Royal Academy of Nutrition Science, National Laboratory of Nutrition Science, and the International Quality & Standard Society, with sponsorship and co-sponsorship by ACPN Canada, ACPN USA, M2 CPN Clinic, Markham CPN Clinic, General Nutrition Group Inc., and other partners. This broad international partnership ensures the document reflects global best practices and is applicable across regulatory contexts.
该技术标准由多家国际领先的科学机构联合制定,包括Keypoint Boston LLC、皇家营养科学院, 国家营养科学实验室、国际质量与标准学会,并由ACPN加拿大、ACPN美国、M2 CPN Clinic、Markham CPN Clinic、General Nutrition Group Inc.等单位赞助和协办。广泛的国际合作确保了文件反映全球最佳实践,并可适用于不同监管体系。
This new standard is now being distributed to natural health product producers, research institutions, and member clinics across ACPN’s network. ACPN formally recognizes this procedure as the authoritative method for confirming the efficacy of natural health products and will encourage its adoption as part of its professional practice guidelines. This will provide consumers, practitioners, and regulatory authorities with more reliable evidence of product benefits, increasing trust and transparency in the marketplace.
目前,该标准已开始向自然健康产品生产商、科研机构以及ACPN网络成员诊所进行分发。ACPN正式认定此程序为确认自然健康产品功效的权威方法,并将推动其作为专业实践指南的一部分。这将为消费者、从业者和监管机构提供更可靠的产品功效证据,提升市场信任度和透明度。
The 78-page document covers the full scope of evidence-based efficacy evaluation, from objectives and scope to data analysis and reporting. Key sections include detailed preparation guidelines for zebrafish, mammalian, and human study platforms, strict adherence to international ethical requirements such as the Declaration of Helsinki, and robust statistical approaches for data processing and validity determination.
该标准全书共78页,全面涵盖了循证功效评估的各个环节,从目标和适用范围到数据分析和报告。核心章节包括:斑马鱼、哺乳动物及人体研究平台的详细准备指南;遵循《赫尔辛基宣言》等国际伦理要求的严格规定;以及用于数据处理和有效性判定的稳健统计方法。

The book provides step-by-step experimental procedures: zebrafish rapid screening for functional efficacy, mammalian model confirmation with CMA-compliant safety testing, and human dietary intervention trials for clinical endpoint validation. It also specifies criteria for informed consent, participant recruitment, and sample size determination, ensuring that human trials are ethically conducted and scientifically robust.
书中提供了分步骤的实验程序:包括用于功能功效快速筛查的斑马鱼实验、符合CMA标准的哺乳动物安全性与功效验证,以及用于临床终点确认的人体膳食干预试验。书中还明确了知情同意、受试者招募及样本量确定的标准,确保人体试验在伦理和科学上均具备严谨性。


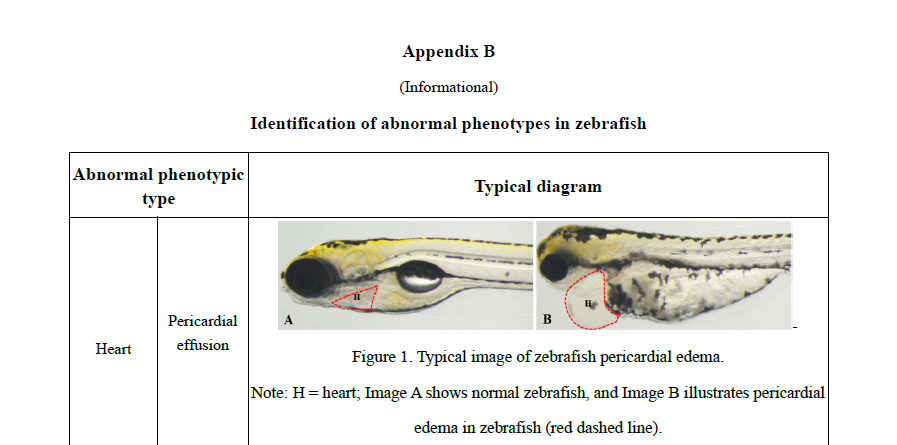
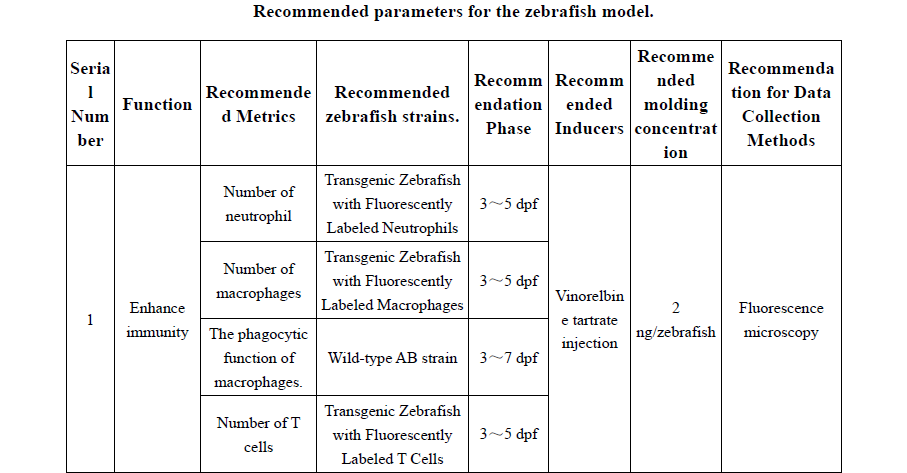
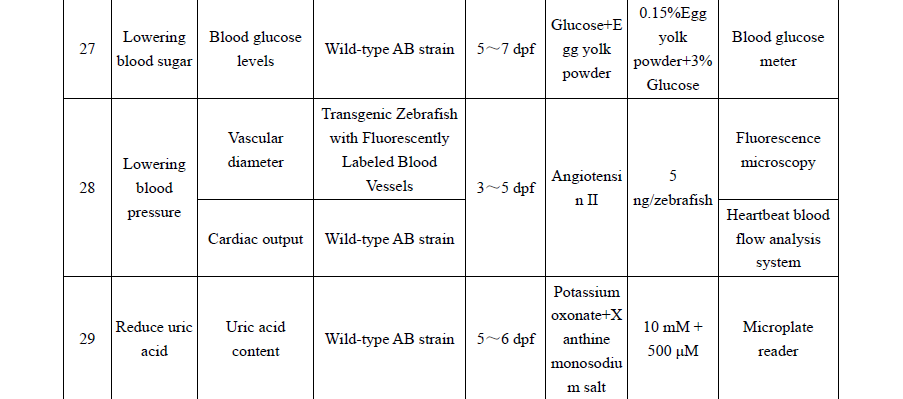
Practical appendices expand the book’s usability, with recommended zebrafish models for 30+ functional outcomes (e.g., immunity enhancement, antioxidant capacity, sleep improvement, lipid-lowering effects), guidance on mammalian pathological models, and phenotypic identification criteria. These tools allow researchers and manufacturers to apply standardized methodologies and improve reproducibility and comparability of results across laboratories.
附录部分进一步提高了该书的实用性,提供了30余种斑马鱼功能模型的推荐参数(如免疫增强、抗氧化、改善睡眠、降脂等),哺乳动物病理模型指南以及表型识别标准。这些工具帮助研究人员和生产商应用标准化方法,提高结果的可重复性和实验室之间的可比性。

By releasing this Technical Standard, ACPN reaffirms its commitment to promoting evidence-based nutrition science and supporting innovation in natural health products. ACPN encourages producers, clinical researchers, and healthcare professionals to adopt these guidelines to ensure the safety, quality, and proven efficacy of products reaching consumers. The distribution of this standard is a milestone toward establishing a transparent and credible system for validating natural health product claims, ultimately benefiting both the industry and public health.
通过发布该技术标准,ACPN再次重申其推动循证营养科学和支持自然健康产品创新的承诺。ACPN鼓励生产商、临床研究人员和健康专业人士采纳该指南,以确保上市产品的安全、质量和功效得到科学验证。本次标准的发布,是朝着建立透明、可信的自然健康产品功效验证体系迈出的重要一步,最终将惠及整个行业及公众健康。